Can New Senolytics Drugs Delay Aging? Part 1: Human Trials Are Coming
The anti-aging race is on, and right now scientists want to know: Can Senolytics drugs delay aging in humans? Seems to work for mice. Human studies are next. Read (and watch) what this could mean to you.
THERE SEEMS to be a race among scientists to discover the first anti-aging drug that successfully passes human trials.
As I’ve written about before, the common diabetic drug, Metformin, and transplant drug, Rapamycin, are already being tested for their promising anti-aging effects (1, 2), and now Senolytics are the darling of the moment.
As CNN reported last week, researchers have turned the spotlight on a new class of drugs that they say could “transform” the field of medicine, as their the first class of drugs developed specifically to treat aging, as if it’s a disease that could be suppressed or eradicated.
This class of drugs are called “Senolytics”.
And the question is:
Can senolytics drugs delay aging in humans?
Researchers from the Mayo Clinic in Rochester, Minnesota aim to move the testing of Senolytic drugs from animal research to human clinical trials. They outlined potential clinical trial scenarios in a paper recently published in the Journal of the American Geriatrics Society.
Said Dr. James Kirkland, director of the Kogod Center on Aging at the Mayo Clinic and lead author of the new paper:
This is one of the most exciting fields in all of medicine or science at the moment.
Read on to learn what discoveries in Senolytics drugs means to how you might age (or not) — and a teaser at the end about how you can get started now with supplements that have Senolytics properties, which I’ll flesh out in Part 2.
In this article, we'll cover:
- Why the connection between cellular apoptosis, senescence and autophagy should matter to you;
- What are Senolytics;
- What mouse studies say about how senolytics drugs delay aging; and
- A very entertaining and informative SciShow video about why we age and what we can do about it.
Let’s dig in…
Can Senolytics Drugs Delay Aging?
 Let’s begin with health and science writer W. Harry Fortuna’s summary of the latest findings into whether senolytics drugs delay aging.
Let’s begin with health and science writer W. Harry Fortuna’s summary of the latest findings into whether senolytics drugs delay aging.
In his article, The first drugs designed to fight aging are ready for human testing published on Quartz.com, Fortuna begins with the “geroscience hypothesis”, which posits that targeting the fundamental mechanisms of aging can help treat or delay the onset of age-related diseases like type 2 diabetes, osteoporosis, and various forms of cancers.
The focus here is to discover ways to increase healthspan — the years in which people are healthy, viable, active members of society – although the underlying fascination is about how these treatments might delay aging itself.
Before I continue with Mr. Fortuna’s article, it would be useful to know the definitions of the following sciency terms, because what happens with them in you could be determinative of your healthspan, if not lifespan:
- Cellular Apoptosis
- Cellular Senescence
- Cellular Authophagy; and
- Senolytics.
Cellular Apoptosis – dead as a door nail
Let’s start with Apoptosis, because unlike the lingering zombish half-death existence of cellular Senescence, or the cellular house cleaning effort of cellular Autophagy, what we have with Apoptosis is death.
Cellular Apoptosis is a process of programmed cell death that occurs in multicellular organisms, among them, we humans.
As Wikpedia puts it:
“Biochemical events lead to characteristic cell changes (morphology) and death. These changes include blebbing, cell shrinkage, nuclear fragmentation, chromatin condensation, chromosomal DNA fragmentation, and global mRNA decay. Between 50 and 70 billion cells die each day due to apoptosis in the average human adult.” (3)
Life and death is normal with biological systems, and thus Apoptosis makes sense. Cellular Senescence, however, is a state of being that keeps surprising those who study it.
Cellular Senescence – when bad things happen to good cells
As (almost famous) anti-aging expert Dr. Judith Campisi and her colleague Dr. Fabrizio d’Adda di Fagagna wrote in Natures Review, our cells constantly experience stress and damage from sources both within (“endogenous”) and external to (“exogenous”) the body.
The cells’ responses from this stress and damage range from complete recovery to cell death, which includes some in-between state whereby the cells are in a “permanent cell-cycle arrest that is termed cellular senescence.”
Drs. Campisi and di Fagangna conclude that:
Understanding the causes and consequences of cellular senescence has provided novel insights into how cells react to stress, especially genotoxic stress, and how this cellular response can affect complex organismal processes such as the development of cancer and ageing. (4)
To put it simply:
Cellular senescence is the phenomenon by which normal cells cease to divide, thereby becoming “senescent”.
Cellular Autophagy – a “self-devouring” process that might extend lifespan
According to Wikipedia, “autophagy” means “self-devouring” in Ancient Greek. In reference to cells this “is the natural, regulated, destructive mechanism of the cell that disassembles unnecessary or dysfunctional components”. (5)
What’s important to know is that cellular autophagy can reduce cellular senescence.
An article published in Nature in 2016 that asserts:
“The regenerative functions of cells decline with age, leading to an irreversible senescence state. Autophagy can keep some of this from happening by reinstating in old cells their former capacity as young cells to maintain proper protein regulation (“proteostasis”), mitochondrial function and oxidative stress repair.”
Translation: Cells that have robust autophagy stay younger!
Senolytics – the senescence destroyer?
Yes, that’s what we want to know: Can Senoytic Drugs instigate Apoptosis in Senescent cells?
Turning again to Wikipedia, a senolytic (from the words “senescence” and “lytic” – destroying) is among the class of small molecules being studied to see if they can selectively induce death of senescent cells; the objective being to delay, prevent, alleviate, or reverse age-related diseases. (6)
Now that you have a sense of what cellular senescence, autophagy and senolytics are about, let’s return to W. Harry Fortuna’s article. He writes that the Mayo Clinic is developing Senolytics as the first class of drugs developed from the ground up to delay or treat aging.
The Senolytics target: Cellular Senescence, a known mechanism of aging
At this point you know that normally cells die a “complete” death known as apoptosis. But that doesn’t always happen, because of that zombified state of Senescence.
The problem with this Senescence is that cells don’t replicate or do anything useful or good, but instead secrete inflammatory chemicals that disrupt the functions of the healthy cells around them.
Scientists aren’t sure why some cells enter Senescence instead of Apoptosis. One theory is that senescence is a defensive response that prevents a cell from becoming tumorous: it halts unchecked growth, stopping a cell from becoming cancer.
Sounds good, but too much of such goodness isn’t.
Concentrations of Senescent cells can cause health problems, too, such as failing heart health, osteoporosis, general run-of-the-mill frailty, and cancer. We don’t want any of this to happen, and that’s why new Senolytic drugs are being designed to target and trigger a specific gene that can ignite Apoptosis in Senescent cells, rendering death upon these zombies.
The Promise of Senolytics Begins With Mice
As usual, it begins with mouse studies.
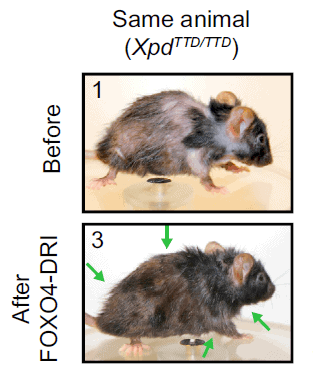
In a study published in 2015 in the journal Aging Cell researchers irradiated mice legs to simulate the ravages of age. So, in effect, the mice legs became physiologically old.
Having prematurely made the mice legs old, the scientists has a moral duty to rejuvenate them, and surprisingly, that’s just what they did.
Just one application of a Senolytic to the affected mice legs reverted their condition to pre-radiation performance, a result that held up over the next seven months. Thus, the Senolytics appeared to reverse the effects of aging!
As earlier mentioned, the scientists involved in such studies are seeking to begin human testing, but they caution that it’s a long and winding round from mice to human studies.
We don’t yet know whether or not there are side effects of Senolytics in humans. There doesn’t seem to be a one-size-fits-all Senolytic for every bodily cell type, so many drugs may need to be developed. Moreover, effectively testing a drug meant to extend the already-long lifespans of humans presents a unique logistical problem — to prove that drugs are preventing people from aging, enough time needs to pass to show that they haven’t.
Despite the inevitable hurdles, the aforementioned lead scientist from the Mayo Clinic study, Dr. James Kirkland, and his team are bullish about the potential for Senolytics to make people healthier and perhaps live longer too boot:
[Once] clinical trials are completed and the potential adverse effects of senolytic drugs are understood fully,” they write, “it is conceivable that the rapidly emerging repertoire of senolytic agents might transform medicine as we know it. (7)
A Quickie on Why We Age and What We Can Do About It
UPDATE (5/7/22): As I explain here, a new study casts shade over the effectiveness of fisetin as a senolytic.
Before I provide a promo on the irrepressible Hank Green’s video rendition about why we age and what we can do about it, here’s a quick look at the forthcoming Part 2 of this two-part series about whether Senolytics drugs delay aging.
Right now, whatever happens with Senolytics drugs is a long way off. Many years will pass before you’ll be able to benefit from them, if ever.
As reported by Healthline, Dr. Kirkland and his Mayo Clinic researchers have already identified several Senolytic drugs that target Senescent cells. A new study published in the journal Nature Communications detailed a screening platform for identifying more drugs targeting these cells, including a new class of drugs called HSP90 inhibitors, including:
- Navitoclax, an experimental anticancer drug;
- Dasatinib, a drug used to fight leukemia;
- Two BCL-XL inhibitors — A1331852 and A1155463;
- Quercetin, a plant polyphenol from the flavonoid group;
- Piperlongumine, a natural product derived from the fruit of the Asian Long pepper; and
- Fisetin, a flavonoid and antioxidant.
It’s odd to me that the last three — Quercetin, Piperlognumine and Fisetin — were lumped with the others and categorized as drugs, because they’re not. These three are widely available supplements, and because they are available now to us, have Senolytics properties, as well as other health benefits, I wonder if they’re worth adding to our supplement regimen now?
That’s the focus of Part 2… does it make sense to begin supplementing with Quercetin, Piperlognumine and Fisetin just in case their Senolytics properties are sufficiently robust to delay aging?
That question will be addressed in Part 2. Until then, watch Hank Green inform and delight in his video below, where he helps us understand the process of aging, informs us of how scientists are studying ways to prevent it and brings us the exciting news of current research in longevity… well, at least for mice.
Click here for Part 2
Last Updated on February 25, 2024 by Joe Garma