Why You Must Detox All The Time, Part 3/3: How To Make Your Detox Pathways Work
Did you know that you have three distinct detox pathways and if one is compromised your toxic load is likely to make you sick and prematurely old? Learn how to make them work and be in harmony.
IN HIS landmark 1947 magnus opus, Detoxification Mechanisms, R.T. Williams defined the field of detoxification. He proposed that non-reactive compounds could be biotransformed in two phases: “Functionalization”, which uses oxygen to form a reactive site, and “Conjugation”, which results in the addition of a water-soluble group to the reactive site.
These two steps — Functionalization and Conjugation, are termed Phase I and Phase II Detoxification. Their aim is to biotransform a lipophilic compound (combines or dissolves in lipids or fats), which is unable to be excreted in urine, into a hydrophilic compound (water-soluble) that can be excreted in urine.
Joining Phase I and II is a relatively recent discovery. Approximately two decades ago, scientists realized that once a toxic substance is made water-soluble it could be excreted out of the cell by means of transport proteins through a process called “Phase III Antiporter”.
Today, detoxification research is ongoing. There are still mysteries to unravel about how the body can deal with such a wide range of toxic compounds it has never encountered, as the world keeps on producing industrial chemicals and heavy metals, pesticides, pharmaceuticals and pathogens – many of which wind up in us.
Part 1 of this three-part series examined why you need to adopt lifestyle habits that enable you to detox all the time, because every day your body is absorbing, inhaling and ingesting exogenous toxins (also refereed to as xenotoxins or exotoxins ) from a toxic world, among them:
- Metabolic end products
- Micro organisms
- Contaminants/pollutants
- Insecticides
- Pesticides
- Food additives
- Drugs
- Alcohol
Part 2 focused on how to fine tune your organs of detoxification; namely your:
- Liver
- Gallbladder
- Gastrointestinal system
- Kidneys
- Lungs
- Skin
- Lymphs
Now here in Part 3 the aim is to get a sense of just how detoxification occurs through the three distinct detox pathways touched upon above, and why they must be fine-tuned and balanced if you’re going to be able to excrete the exogenous toxins that day by day enter your body and demand a tax be paid in the currency of health.
In this article you'll discover:
- How each of the three phases of detoxification work alone and together;
- What induces or inhibits each phase’s activity;
- What regulates these detoxification pathways; and
- Why it’s vitally important that your detoxification pathways are tuned and in harmony.
Before we start, a shout out to Dr. DeAnn J. Liska, author of The Detoxification Enzyme Systems, Herbal Transitions’ Biotransformation Pathways and Carahealth’s Phase 1 and 2 Detoxification Pathways – all of which ably served as the scientific resources for much of this article.
The Enzymes of Detoxification
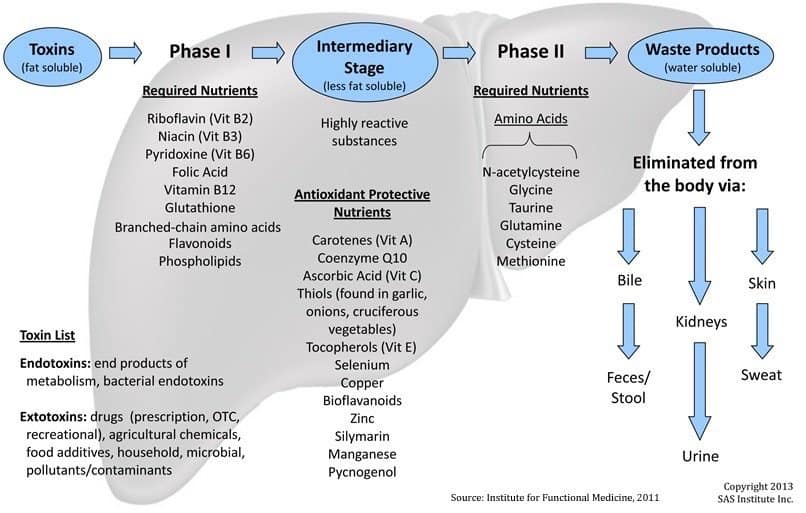
There are three detox pathways or phases of detoxification where enzymes rule the roost. This illustration tells the story at a glance:
The rest of this section spells it out.
Phase I Detoxification: Enzymatic Biotransformation
Phase I detoxification is often referred to as “Enzymatic Biotransformation”. Biotransformation is the process whereby chemicals, such as toxins, are processed by the body’s enzymes to make them different, less harmful or able to be more rapidly excreted.
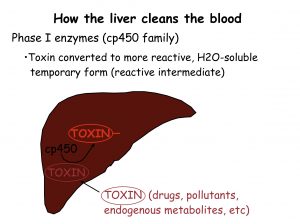
In this first of three phases of detoxification, a supergene family of specialized enzymes called Cytochrome P450s (“CYP450”) transform lipid-based toxins (which most of them are) into hydrophilic toxins. As mentioned, once their hydrophilicity is increased, the toxins can be further processed by the kidneys and eliminated via urine. (As you’ll learn later, Phase II further increases hydrophilicity through a process called “conjugation”.)
In a typical Phase I reaction to a toxin, a CYP450 enzyme uses oxygen and NADH (a coenzyme found in all living cells) as a cofactor to add a reactive group, such as a hydroxyl radical.
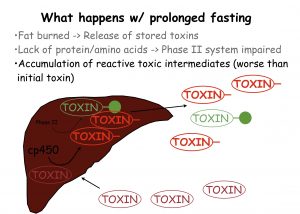
This example means nothing to you if you’re not well versed in biochemistry, but what is meaningful is to know that as a consequence of this step in detoxification, reactive molecules are produced (such as free radicals) that may be more toxic than the “parent” molecule was at the start. If not further metabolized by Phase II conjugation they may cause damage to proteins, RNA, and DNA within the cell.
The CYP450 enzymes employed in Phase I are the first enzymatic defense that acts to metabolize exogenous toxins, including pharmaceuticals, and many are neutralized here. Others are transformed into intermediate forms that are then processed by Phase II. These intermediate forms are often more chemically active than they originally were and thus can be more toxic. If the Phase II detoxification systems are not working adequately, these intermediates can cause substantial damage, including the initiation of carcinogenic processes or genetic mutations and the creation of free radicals.
Free radicals are a significant side effect of Phase I activity. For each molecule of a toxin metabolized by Phase I, one molecule of free radical is generated. Without adequate free radical defenses, every time the liver neutralizes a toxin, it’s damaged by the free radicals produced.
You may have heard of free radicals before, given that many supplements you may use are made from antioxidant substances thought to reduce free radical damage. Free radicals are atoms or molecules with an odd (unpaired) number of electrons that cause damaging chain reactions to cellular components such as DNA, or the cell membrane, potentially causing cellular dysfunction or death.
The good news is that antioxidants like glutathione are molecules that can safely interact with free radicals and terminate the chain reaction before vital molecules are damaged. Of course for this to happen, a sufficient amount of antioxidants to deal with the toxic load must be present in the body, otherwise the original toxins can become more deadly (those so-called “intermediates”) than they were at the start of the Phase I process, as mentioned.
The common spice turmeric, found most commonly in curry powder, slows down Phase I while egging on phase II, an effect that can help to move toxins more quickly through the two phases. This may explain turmeric’s powerful anticancer properties.
Certain nutrients act as coenzymes to the Phase I detoxifying enzymes. Vitamins B2, B3, B6, and B12 plus folic acid, bioflavonoids, branched-chain amino acids, and phospholipids are needed to keep Phase I going smoothly.
It is possible to calculate the functioning level of Phase I detoxification by undertaking an easy caffeine metabolism examination. This test involves ingesting a known amount of caffeine and taking samples of saliva twice at particular intervals.
There is a direct relation between the effectiveness of caffeine clearance and Phase I detoxification competence. Rapid clean up of caffeine demonstrated increased production of detoxification enzymes either owing to exposure to xenobiotics or the body’s internal toxins. On the other hand, a sluggish clearance of caffeine is an indication of the fact that there is an abnormal activity of the enzyme cytochrome P-450 within the liver.
People having sluggish caffeine clean up usually face problems in getting rid of xenobiotics as well as other toxic substances from the body. In the absence of a saliva test, you can get some sense of your PI detox effectiveness by noting how quickly you clear the effects of caffeine or whether it makes you sweat, get nervous or jittery. The less you’re affected by caffeine, the better your P1 is working.
Phase II Detoxification: Enzymatic Conjugation
Phase II detoxification is often referred to at the “conjugation pathway”. From the perspective of biology, to “conjugate” is to become temporarily united in order to exchange genetic material.
In Phase II, liver cells add or unite (conjugate) other substances (such as cysteine, glycine or a sulfur molecule) to a toxic chemical or drug. This happens either directly, or after first being activated by Phase I enzymes.
As with Phase I, the objective here is to chemically change toxins into water-soluble compounds that can be eliminated from the body by the kidneys, or through the bile should the chemical have an elevated molecular weight (a heavier molecular mass).
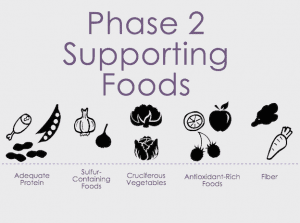
This chemical biotransformation is achieved by one or more of seven distinct major biochemical reactions (often referred to as “pathways”), each requiring specific nutrients to conjugate with in order to excrete specific types of toxins. The seven pathways are glutathione conjugation, amino acid conjugation, methylation, sulfation, acetylation, glucuronidation, and sulfoxidation. These reactions require cofactors that must be replenished through dietary nutritional sources. (A “cofactor” is a substance whose presence is essential for the activity of an enzyme.)
The nutrients required for Phase II fall into two categories. The first are the amino acids, which donate molecules that are attached to Phase I intermediates. These include the sulfur donors, among which are the amino acids methionine, taurine, cysteine, and N-acetylcysteine. Other, non-sulfur-containing amino acids are also required: glycine, taurine, glutamine, ornithine, and arginine. The antioxidant amino acid glutathione is also required for Phase II detoxification.
A diet low in protein-all too common in people trying to lose weight with a low-fat diet-can dramatically slow Phase II detoxification. Aspirin and other non-steroidal anti-inflammatory drugs (“NSAIDs”), including ibuprofen (found in Advil, Nuprin, and other products), also slow phase II detoxification.
Interestingly, most Phase II enzymes are controlled at the genetic level by a protein called nuclear factor erythroid-derived 2 (“Nrf2”), a master regulator of antioxidant response.
Nrf2 “senses” oxidative stress or the presence of toxins in the cell, and allows the cell to mount an appropriate response, typically through the synthesis and activation of important detoxification molecules including superoxide dismutase (“SOD”), as well as glutathione and coenzyme Q10 (“CoQ10”), two potent antioxidants.
This is worth noting because cellular damage done by free radicals is significant enough to have inspired the Free-radical Theory of Aging, which hypothesizes that organisms age because cells accumulate free radical damage over time resulting in specific changes to cell biology that contribute to the aging process. If therefore, Nrf2 activation can be optimized during Phase II biotransformation, then the robustness of the body’s detoxification system plays a key role in the slowing down the degradation associated with aging.
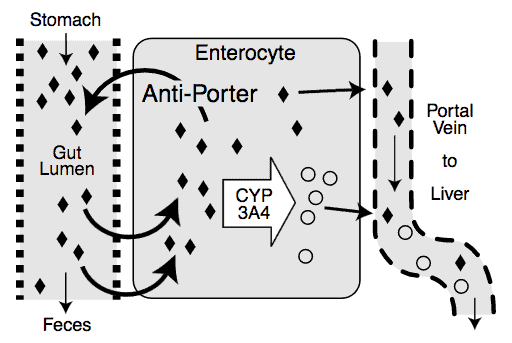
Phase III Detoxification: Transport, The Antiporter System
Most of the literature on detoxification focuses on Phase I and II and the dominant roles that the kidneys and liver have in those processes. It’s now understood that the gut also plays an important detoxification role, which is unsurprising. It is in the small intestine of the gut where the first evidence of xenobiotic exposure occurs and, over the course of a lifetime, it is here where the largest load of antigens and xenobiotics confront the human body.
Over the last 20 years, or so, scientist have been trying to establish the extent to which an additional detoxification process is occurring in the small intestine known as “antiporter activity”.
What’s antiporter activity, you ask?
The antiporter is an energy-dependent efflux pump (a cellular pump), which pumps exogenous toxins out of a cell, thereby decreasing the intracellular concentration of them.
Sorry you asked, right?
Let’s just accept that airporter activity is an important factor in the first pass metabolism of pharmaceuticals and exogenous toxins. The current consensus is that pumping non-metabolized xenobiotics out of the cell and back into the intestinal lumen may allow more opportunities for Phase I activity to metabolize the xenobiotic before it’s taken into circulation throughout the rest of the body.
In addition to the gut, Phase III transporters are present in many tissues, including the liver, kidneys, and brain, where they can provide a barrier against xenobiotic entry, or a be the mechanism for actively moving xenobiotics and endobiotics in and out of cells.
Since water-soluble compounds require specific transporters to move in and out of cells, Phase III transporters are necessary to excrete the newly formed Phase II products out of the cell.
In the liver, Phase III transporters move glutathione, sulfate, and glucuronide conjugates out of cells into the bile for elimination. In the kidney and intestine, phase III transporters can remove xenobiotics from the blood for excretion from the body.
Once an unwanted compound has been completely biotransformed and removed from the cell, it will then be eliminated from the body via – kidneys, bowels, breath, sweat, saliva or hair – completing the detoxification process.
Detox Regulation
Since the detoxification systems function to help in the management of exposure to exogenous compounds, the body has developed several mechanisms to regulate detoxification activity.
Specific detoxification pathways may be induced (enhanced) or inhibited (repressed) depending on the presence of various dietary or xenobiotic compounds, the age and sex of the individual, genetics, and lifestyle habits, such as smoking.
Furthermore, disease can also influence activity of the enzymes. In some disease states, detoxification activities appear to be induced or up-regulated, whereas, in other conditions these activities may be inhibited from acting or not produced at high levels.
Induction Enhances Detoxification
When the body is confronted with a high xenobiotic load, the Phase I and/or Phase II enzymes involved in detoxifying it can be induced, leading to more enzymes being present and a faster rate of xenobiotic detoxification.
Inducers can be mono-functional or multi-functional.
As the names suggest, a mono-functional inducer affects only one enzyme or one phase of detoxification; whereas a multi-functional inducer affects multiple activities.
Mono-functional inducers, such as polycyclic hydrocarbons from cigarette smoke and aryl amines from charbroiled meat, result in dramatic induction of specific enzymes, leading to a substantial increase in Phase I activity, with little or no induction of Phase II enzymes.
That’s a problem.
Induction of these activities without co-induction of Phase II activities may lead to an uncoupling of the Phase I and Phase II balance of activity and, thereby, a higher level of reactive intermediates mentioned above, which can cause damage to DNA, RNA, and proteins.
The multi-functional inducers include many of the flavonoid molecules found in fruits and vegetables. For example, ellagic acid found in red grape skin has been shown to induce several Phase II enzymes while decreasing Phase I activity.
Garlic oil, rosemary, soy, cabbage, and brussels sprouts all contain compounds that can induce several Phase II enzyme activities.
Commonly, the glutathione S-transferase and glucuronyl transferases are induced by multi-functional inducers. In general, this increase in Phase II supports better detoxification in an individual and helps to promote and maintain a healthy balance between Phase I and Phase II activities.
The enhancement of Phase II activity has been proposed to explain, at least in part, the ability of fruits and vegetables to protect against many cancers.
Inhibition Suppresses Detoxification
Phase I and Phase II enzyme activities can also be inhibited. Inhibition can occur between two or more compounds competing for the same detoxifying enzyme, or by an increase in toxic load that overwhelms the system and its detoxification enzyme activities.
What can also lead to inhibition are compounds that selectively inhibit only one detoxifying activity, such as:
- Quindine, a drug used to treat irregular heart beats, competitively inhibits the activity of a particular Phase I enzyme, CYP2D6.
- Cimetidine, a drug used to treat ulcers and gastroesophageal reflux disease (“GERD”), is an example of a compound that can bind directly to the heme iron of the CYP450 reactive site to inhibit all cytochrome-dependent Phase I enzyme activities, which could essentially switch off Phase I.
A more general inhibitor is grapefruit juice. It contains high amounts of the flavonoid naringenin which allows it to inhibit first pass metabolism of many drugs that are detoxified through the CYP3A4 enzyme-antiporter system in the intestine.
Polymorphisms Affect Detox Regulation
“Polymorphisms” refers to the occurrence of different forms or genetic variations among members of a population. Genetic differences in the ability of an individual to metabolize xenobiotics are related to the presence of different versions of the gene encoding that activity, or genetic polymorphism.
To give you a sense of this, let’s consider CYP2D6, a classic example of the influence of genetic polymorphism on phenotype.
Several varieties of the CYP2D6 gene exist in the population; some encode an enzyme with a lower activity than others. Individuals who receive two versions of the gene encoding slower CYP2D6 activity do not detoxify substances through the CYP2D6 pathway as fast as those who receive genes encoding faster acting enzymes.
These individuals have been termed “slow metabolizers”.
The CYP2D6 enzyme is an important detoxifier of many narrow spectrum drugs, including antiarrhythmics, antidepressants, and antipsychotic drugs. Adverse side effects occurring from these drugs may be reduced by decreasing dosages in those individuals who are CYP2D6 slow metabolizers.
Other Factors Regulate Detoxification
Various factors affect influence the expression and resultant activity of many detoxification enzymes, such as sex and age. The CYP3A family of detoxification enzymes is particularly sensitive to hormones, such as progesterone, which may regulate it. Premenopausal women generally show 30 to 40% more CYP3A4 activity than men or postmenopausal women.
Since CYP3A4 is the primary Phase I detoxification pathway for the anti-epileptic agents phenobarbital and phenytoin, pregnant women, in whom this activity is increased, more readily eliminate these drugs and consequently may require a higher dose during pregnancy.
Disease and health status of the individual also influence detoxification activities. Since the majority of detoxification occurs in the liver, impairment of normal liver function due to alcoholic disease, fatty liver disease, biliary cirrhosis, and hepatocarcinomas can lead to lower detoxification activity.
These are just a few examples of the many numerous ways different enzymes and genes mutually affect each other leading to different detoxification outcomes.
Your Takeaway
If your three detox pathways are not functioning in harmony with each other, the toxins that come into your body on a daily basis will accumulate until you get sick and prematurely old.
Although it’s smart and effective to set aside a block of time several times a year to focus on tweaking your diet and supplementation to aide your detox processes, doing so will not suffice if the rest of the year is spent without adequate nutrition required by your Phase I, II and III detoxification pathways.
At minimum, ensure you consume plenty of fiber, lean protein, omega-3 fatty acids, glutathione (in liposomal form is best), N-acetyl cysteine, alpha lipoic acid, B-complex, curcumin or turmeric, sulphurous vegetables (artichoke, asparagus, bean sprouts), cruciferous vegetables (broccoli, cabbage, Brussel sprouts, bok choy), green leafy vegetables (kale, spinach, chard) and dark thin-skin fruits (black berries, blue berries).
Just scroll back up to that first illustration and take note of the antioxidants and nutrition there listed.
P.S. If you’d like to get a sense of different detox protocols, supplements, etc., check out my various articles on the subject.
Last Updated on February 27, 2022 by Joe Garma