Time Restricted Feeding Improves Metabolic Health and More
Time restricted feeding improves the metabolic health in mice, and likely in you too. The reason is circadian rhythms. Eat when you’re most metabolically active. And that’s during the day, not at night.
There is an ongoing tug of war regarding the health benefits of Time Restricted Feeding (TRF) on metabolic health. It appears to me that this argument is not centered on the same set of conditions; meaning, that various studies that appear to come to contradictory conclusions are not measuring the same thing, or are not studying the same cohorts.
Oftentimes the results of a TRE study conflict with that of another seemingly similar study, but:
- One study’s cohort is obese people, the other is not.
- One study’s cohort is old, the other is young.
- One study’s feeding window is different than the other (smaller/larger, in the morning, mid day, or evening).
- One study’s cohort has more of one gender than another (males and females don’t always have the same result).
- In some studies, one group loses weight on TRE, not in the other; however, the calories might not have been identically restricted, or the type of food was different.
I’m going to attempt to straighten some of this out, notwithstanding the fact that the impact of Time Restricted Feeding on metabolic health is not settled science.
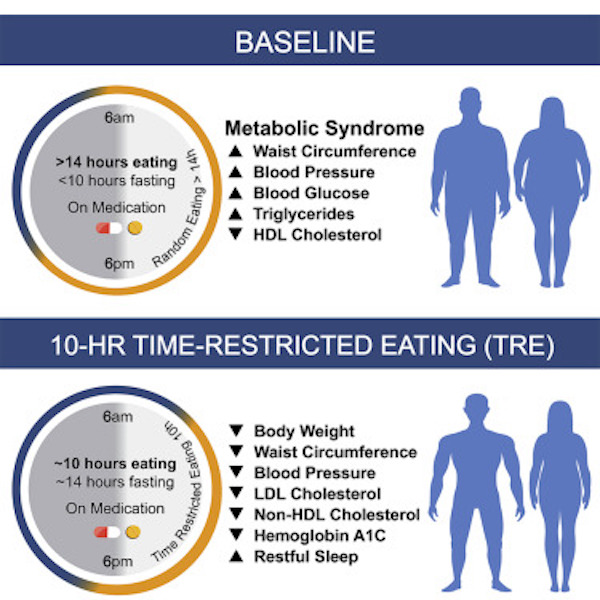
Also, we’re going to stray father afield than just looking at “metabolic health”, which can be defined by having blood sugar, waist circumference, blood pressure, cholesterol, and triglycerides all within a healthy range, a benchmark set by scientists and doctors. Some other metrics that will be examined include gene expression in several organs.
If you don’t want to wade into the particulars, go to Your Takeaway at the end of this post to get the low down.
Let’s dive in…
The Health Benefits of TRF
 A 2023 study with the ponderous title, Diurnal transcriptome landscape of a multi-tissue response to time-restricted feeding in mammals, studied the impact of Time Restricted Feeding on the metabolic health of mice, as well as some other health parameters.
A 2023 study with the ponderous title, Diurnal transcriptome landscape of a multi-tissue response to time-restricted feeding in mammals, studied the impact of Time Restricted Feeding on the metabolic health of mice, as well as some other health parameters.
Humans are not mice, but there are many biological similarities that make mice the cohort that’s often studied as a preliminary test to determine if something is worth pursing in humans, if possible.
This post will review this study, which was led by the prominent scientist, Professor Satchidananda Panda of the Salk Institute. His work explores the genes, molecules and cells that keep the whole body on the same circadian clock.
The circadian clock has an internally driven 24-hour rhythm that resets every day by the sun’s light/dark cycle. It has an important purpose: It prepares your body for expected changes in the environment, such as the time for activity, time for sleep and times to eat.
Dr. Panda’s research was first to discover that mice who eat within a set amount of time (8-12 hours) are slimmer and healthier than those who ate the same number of calories during a larger window of time (in excess of 12 hours per day). If this holds true in humans, it could have profound impacts on treating overeating disorders, diabetes and obesity.
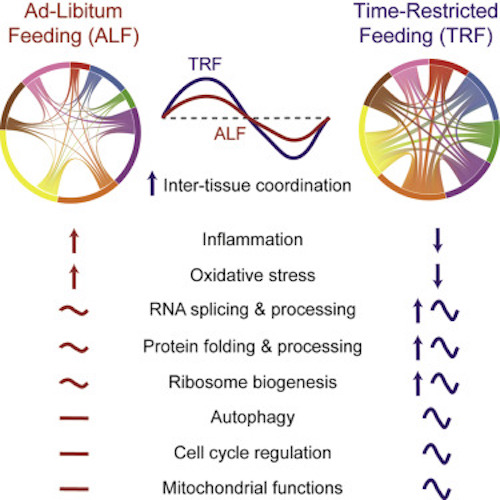
Panda’s 2023 study of mice showed that Time Restricted Feeding “TRF” profoundly impacts gene expression — nearly 80% of all genes showed differential expression or rhythmicity under TRF in at least one tissue.
TRF, TRE and Intermittent Fasting (IF) are often used interchangeably. TRF is most often used with animal cohorts, TRE with human cohorts, and IF can refer to various fasting periods within a day or week. Gene expression refers to genes that are "turned on", or expressed physiologically. Rhythmicity or "biological rhythm" are often used interchangeably with circadian rhythm, bodily functions regulated by your internal clock, such as sleep and wakefulness, body temperature, hormone secretion, etc.
How the study was conducted
There were two groups of mice:
- Group 1 ate whenever they wanted (“ALF” ad libitum feeding).
- Group 2 were fed on a TRF schedule that I couldn’t find in the study.
- Both groups were fed a “western diet”, considered obesogenic (causing obesity).
- In both groups, the gene expression changes in samples were taken from 22 organs and brain regions collected every two hours over a 24-hour period.
- The experiment lasted for seven weeks.
Results of the study
These data indicate that TRF prevented high fast diet-induced physiological defects in multiple tissues.
- The two groups consumed the same amount of obesogenic food, causing the ALF mice to gain weight, adiposity (fat), and metabolic dysfunction; whereas TRF mice did not.
- The TRF group had a greater synchronized and shared rhythmic gene expression across tissues, which is known to improve physiology.
- TRF affected gene expression in major metabolic organs: Various biomarkers in the livers, skeletal muscles, white adipose tissue, gut and heart of the TRF group were all improved.
Click here to read the particulars
In Liver:
TRF upregulated pathways were involved in complement proteins, IGF1 signaling, and protein glycosylation and increased rhythmicity of genes involved in ketone body and one carbon metabolism, cholesterol and bile acid metabolism, and very-low-density lipoprotein (VLDL) particle assembly, while downregulated pathways were involved in glucose metabolism (gluconeogenesis and the pentose phosphate pathway) and glycerolipid metabolism.
In skeletal muscle:
TRF upregulated pathways in glucose metabolism (glycolysis and pyruvate metabolism), mitochondria organization, and muscle regeneration; downregulated pathways were hypoxia inducible factor-1 (HIF-1), tumor necrosis factor (TNF), and growth factor signaling.
In eWAT:
[In Epididymal white adipose tissue (eWAT) secretes an array of cytokines to regulate the metabolism of organs and tissues in high-fat diet (HFD)-induced obesity, but its effects on bone metabolism are not well understood.]TRF upregulated pathways were involved in glucose metabolism (glycolysis and pyruvate metabolism), fatty acid metabolism, BCAA catabolism, tricarboxylic acid (TCA) cycle, oxidative phosphorylation, and mitochondria organization, while downregulated pathways were involved in immune activation, cell cycle regulation, and inflammatory signaling.
In the gut:
TRF seemed to dampen inflammation by decreasing nuclear factor-kB (NF-kB), toll-like receptor (TLR), and IL-17 signaling in the duodenum, jejunum, and ileum, while increasing the expression and rhythmicity of genes involved in glucose, lipid, and xenobiotic metabolism, as well as cell cycle regulation.
In the heart:
TRF (1) improved mitochondria organization, nitric oxide (NO) signaling, and glucose metabolism; (2) induced rhythmicity in pathways regulating cytoskeleton organization and muscle differentiation; and (3) inhibited TLR signaling, c-Jun N-terminal kinase (JNK) signaling, and succinate metabolism. In the KIC (kidney cortex) and KIM (kidney medulla), TRF improved rhythmicity in fatty acid catabolism, ketone body metabolism, and mitochondria organization while suppressing xenobiotic and nucleotide metabolism, as well as JNK (c-Jun N-terminal kinase) and Rho GTPase signaling.
In BAT (brown adipose tissue):
TRF increased rhythmicity in autophagy, adipogenesis, and thermogenesis while decreasing cell cycle processes and TNF signaling.
Study Conclusions and Limitations
The study makes these two important conclusions:
(1) The timing of feeding-fasting cycles (TRF) regulates the expression of several tissue-specific transcription factors, which, in combination with clock proteins, can drive rhythmic gene expression.
Transcription factors are proteins involved in the process of converting, or transcribing, DNA into RNA, which is the process by which the information encoded in a gene is turned into a function. Clock proteins are controlled by "clock genes". The levels of these proteins rise and fall in rhythmic patterns, and control various functions, including when we sleep and rest, and when we are awake and active.
(2) Although consumption of a high fat and high carbohydrate diet is associated with poor health outcomes, including reduced healthspan and lifespan, several hallmarks of aging were reversed by TRF, resulting in reduced levels of inflammation, increased autophagy, improved RNA and protein homeostasis, and augmented metabolic flux. (Metabolic fluxes are the significant driving force in cell physiology, as they determine the proportion of various pathways in all cellular functions and metabolic processes.)
Limitations cited by the study include:
- The mouse cohort was young males. Future studies will be needed to understand how TRF-dependent gene expression changes across tissues are affected by sex and age, especially since TRF is known to affect physiology in both age- and sex-dependent and -independent manner.
- The study could not distinguish between gene expression that is causal vs. consequential as an effect of TRF.
- Cell-type-specific effects could not be discerned. For example, the effect of TRF on immune functions in eWAT (epididymal white adipose tissue) and iWAT (inguinal white adipose tissue) could be identified, but not the specific immune cell type driving this phenotype.
If you have the time, check out Dr. Rangan Chatterjee’s interview of Dr. Panda about TRF:
I’ve also written two posts about Dr. Panda’s work:
Sync Your Circadian Rhythms and Time Restricted Feeding to Optimize Health
When You Eat Is More Important Than What You Eat, Says Dr. Panda
Your Takeaway
Remember these four things:
- Even though, presumably, you’re not a mouse, research on mice are conducted to determine if a particular hypothesis is worthy of being adjudicated in human trials; therefore, the results of such experiments may be relevant to humans.
- When you eat matters, perhaps more than what you eat. You want to eat your food when you’re most metabolically active, which for a human is during the day. In the mouse study reviewed above, even though both groups of mice ate the same amount of fattening food, only the ones who ate whenever they wanted got fatter and unhealthy. (Humans tend to eat for 15 hours each day — that’s a problem.)
- Mice are nocturnal, and so eating at night is normal for them. Not for you. Don’t eat late at night. Doing so disrupts your metabolism and encourages weight gain.
- Try eating within an eight-hour window that ends three to four hours before bedtime, such as between 11:00 AM and 7:00 PM.
If you have any questions or comments, please scroll down and type them into the Comments section,
Last Updated on July 7, 2023 by Joe Garma