Can Aging Hallmarks Predict Age-related Disease?
Specific aging hallmarks may be able to predict many age-related diseases. If you can identify which age-related disease you’re likely to get based upon your genetics or lifestyle factors, you can identify the therapeutic intervention now.
 The fast answer to the question, Can aging hallmarks predict age-related disease is “yes”. How this happens is quite remarkable. The story needs to begin with a much-cited scientific paper, the Hallmarks of Aging, and then pivot to one published in 2020 that used the “Hallmarks” revelations to determine how they are associated with the many age-related diseases that debilitate nearly everyone as they get older.
The fast answer to the question, Can aging hallmarks predict age-related disease is “yes”. How this happens is quite remarkable. The story needs to begin with a much-cited scientific paper, the Hallmarks of Aging, and then pivot to one published in 2020 that used the “Hallmarks” revelations to determine how they are associated with the many age-related diseases that debilitate nearly everyone as they get older.
In 2013 Dr. Carlos López-Otín and fellow researchers published a paper called the Hallmarks of Aging that has become the gold standard for explaining how aging happens. There have been challenges to some of the assertions made in that paper, some of which I’ll mention later, but the nine Hallmarks have largely stood the test of time, and so far remains the most common bedrock explanation about how we age from which many subsequent studies rely.
Why should this matter to you?
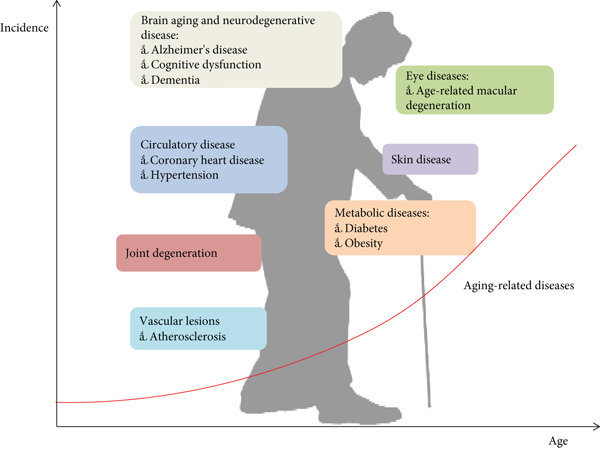
It matters, because these hallmarks are thought to be fundamental biological mechanisms shared across multiple major age-related diseases such as cancer, diabetes, cardiovascular disorders and neurodegenerative diseases. If you can know the aging factors that are strongly associated with a major disease that you have a good chance of getting, it would be helpful to search for the appropriate therapy now.
So, let’s dig in and find out.
Content
- The 9 Hallmarks of Aging
- Primary
- Antagonistic
- Integrative
- Criticism of
- Age related diseases and the Hallmarks
- Five Hallmark aging mechanisms and ARDs
- Interventions that target aging Hallmarks
- Senolytics and rapamycin
- Interventions that may inhibit mTOR
- Your Takeaway (if you’re in a rush)
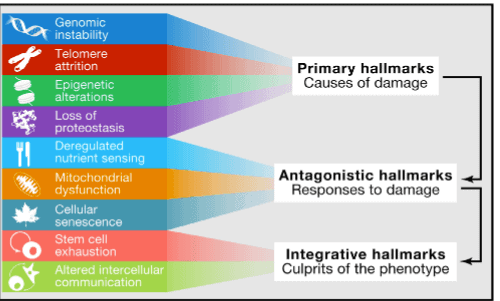
The 9 Hallmarks of Aging: Primary, Antagonistic and Integrative
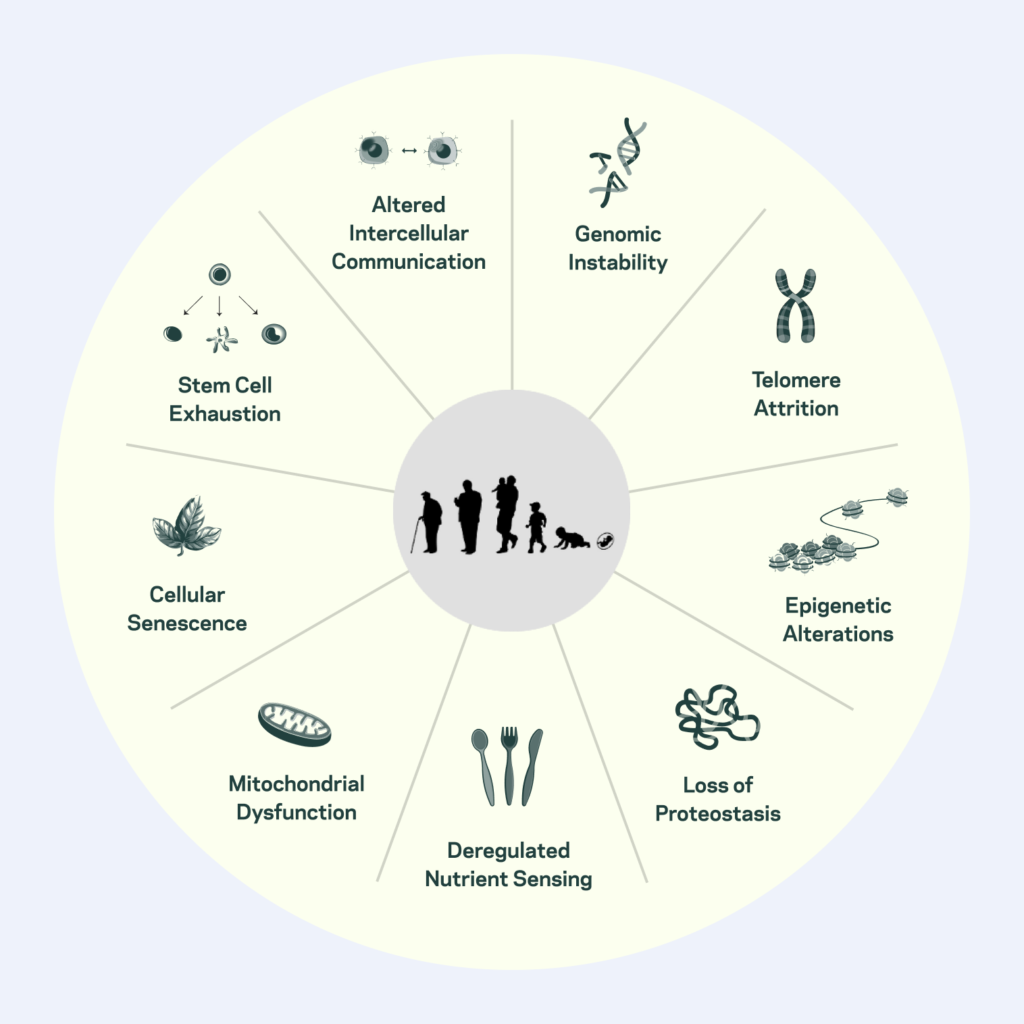
The ¨Hallmarks of Aging¨ describe the basic processes thought to underlie aging in different organisms. These hallmarks are thought to be fundamental mechanisms shared across multiple major age-related diseases such as cancer, diabetes, cardiovascular disorders and neurodegenerative diseases.
The nine Hallmarks of Aging are:
- Genomic instability: During aging, our DNA becomes damaged, especially in stem cells and fast-dividing cells. Damaged DNA may result in improper cell function which impacts proper functioning of tissues and organs.
- Telomere attrition: As we age, the ends of chromosomes, known as telomeres, get shorter and cannot protect DNA any longer. This can increase the likelihood of genomic instability and impair cell function.
- Epigenetic alterations: During aging, the epigenome — the molecular machinery that regulates our genes’ activity — becomes disorganized. This leads to certain beneficial genes being turned off when they should be on, and genes that can cause trouble being turned on when they should be off.
- Loss of proteostasis: Over time, more and more proteins accumulate inside and outside our cells. These proteins should be cleaned up, and when they aren’t, interfere with the cells’ proper functioning.
- Deregulated nutrient-sensing: During aging, our cells become less tuned to nutrient signals, which disrupts a cell’s ability to utilize and produce energy. This can result in reduced energy and metabolic dysfunction.
- Mitochondrial dysfunction: During aging, mitochondria — the power plants of our cells — become dysfunctional. Without adequate power, cells — and by extension, organs — are not able to perform as well as when they were young.
- Cellular senescence: Senescent cells are former healthy cells that stopped dividing and secrete substances that damage healthy surrounding cells. Commonly referred to as “zombie cells,” these cells increase exponentially with age and lead to tissue and organ damage.
- Stem cell exhaustion: With age, stem cells become dysfunctional or die off. Since stem cells are responsible for producing new copies of our cells when they’re needed, having fewer or dysfunctional stem cells leads to our tissues being less replenished & maintained.
- Altered intercellular communication: When we get older, our cells become increasingly exposed to a hostile environment, characterized by pro-inflammatory and other deleterious substances that hinder cells, including stem cells, to function properly.
Thanks to Novos Labs for brief summaries of Hallmarks. For more detailed descriptions go to The Reasons We Age and What To Do About It.
These Hallmarks are grouped into three main categories according to their functional hierarchy:
Primary Hallmarks
- Genomic instability
- Telomere attrition
- Epigenetic alterations
- Loss of Proteostasis
Antagonist Hallmarks
The antagonistic hallmarks of aging are Hallmarks that can have beneficial or deleterious effects on the cell, depending on the level of intensity. As such, they are considered to be traits resulting from compensatory responses to damage to the manifestation of primary hallmarks, which have varying effects on health depending on the intensity of their activity: low levels of cellular senescence, for instance, may confer health benefits, whereas, alternatively, high levels of cellular senescence may do the opposite and cause accelerated aging. Antagonistic hallmarks are:
- Deregulated nutrient sensing
- Mitochondrial dysfunction
- Cellular senescence
Integrative Hallmarks
Integrative hallmarks of aging are hallmarks that have a more direct effect on the tissue homeostasis and function. They are the dysfunctional damage resulting from the previous two groups of hallmarks that lead to further operational deterioration associated with aging. Integrative hallmarks are:
- Stem cell exhaustion
- Altered intercellular communication
To sum it up: (1) Issues with our genes, telomeres, epigenetics, and proteostasis create the damage associated with aging; (2) In response to that damage, nutrient sensing pathways and mitochondria functionality get impaired, and cells become senescent; and (3) All this damage results in stem cell exhaustion and altered intercellular communication.
Criticism of the Hallmarks of Aging
A definitive, tested, agreed upon uniform theory of aging does not yet exist. Every current theory can be picked apart to some degree, and so it is with the Hallmarks of Aging. Although each of the nine Hallmarks are thought to contribute to the aging process, definitive evidence is missing.
Scientists have yet to confirm that ameliorating these Hallmarks would result in a decelerated aging phenotype and lifespan extension, thus providing evidence that they are upstream of the aging process. Also, some studies suggest that aging is largely regulated in a tissue-specific manner and that age-dependent changes vary enormously according to cell type.
You may read a critical review of the Hallmarks of Aging here.
Age-related Diseases and Hallmarks of Aging
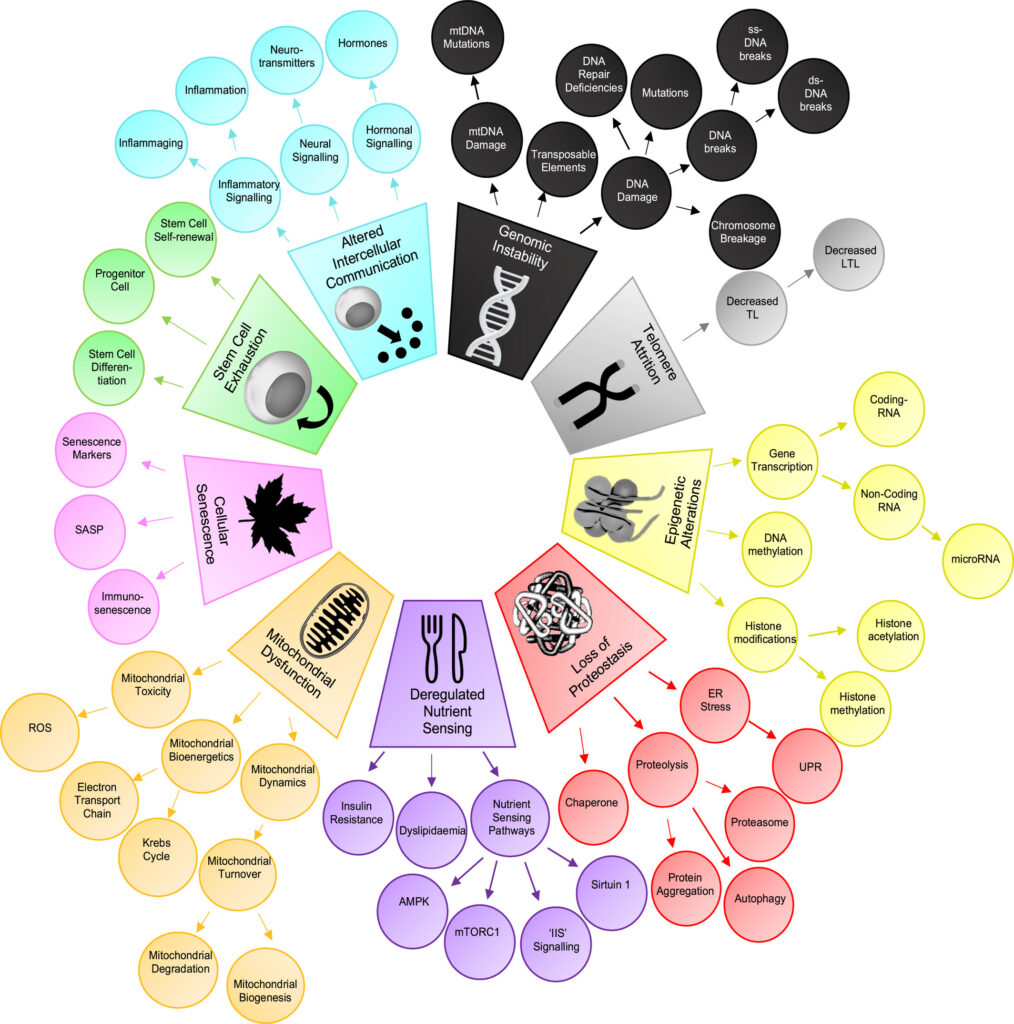
The “Hallmarks of Aging” expanded into a taxonomy. The nine original aging hallmarks were expanded into a taxonomy of 65 related terms and four levels. (Click to enlarge.)
In a paper entitled Biological mechanisms of aging predict age-related disease co-occurrence in patients published in May, 2020, researchers connected the dots between five of the nine Hallmarks of Aging and chronic disease.
This connection answers why you should care about the Hallmarks to begin with: They induce the downstream effect of enabling many of the chronic diseases that become more and more prevalent as we age:
“… individual mechanisms of aging might contribute to the co-occurrence of age-related diseases in humans and could be targeted to prevent these conditions simultaneously.”
Researchers conducting the study text mined 917,645 literature abstracts followed by manual curation and found strong, non-random associations between age-related diseases and aging mechanisms in humans. These associations were integrated with clinical data from 3.01 million patients and showed that 30 top age-related diseases associated with each of five aging mechanisms were much more likely than chance to be present together in patients.
The 5 Hallmark Aging Mechanisms and ARDs
These are the five aging mechanisms the study determined predicted most of the downstream age-related diseases:
- Deregulated nutrient sensing
- Mitochondrial dysfunction
- Cellular senescence
- Stem cell exhaustion
- Altered intercellular communication
Note that the first three of the five Hallmarks associated with age-related diseases are Antagonistic Hallmarks (compensatory responses to damage), and the last two are Integrative Hallmarks (dysfunctional damage).
The specific age-related diseases (ARDs) associated with the Hallmarks were extensively researched, but are difficult to determine by the study’s graphical depictions; for instance, this one:
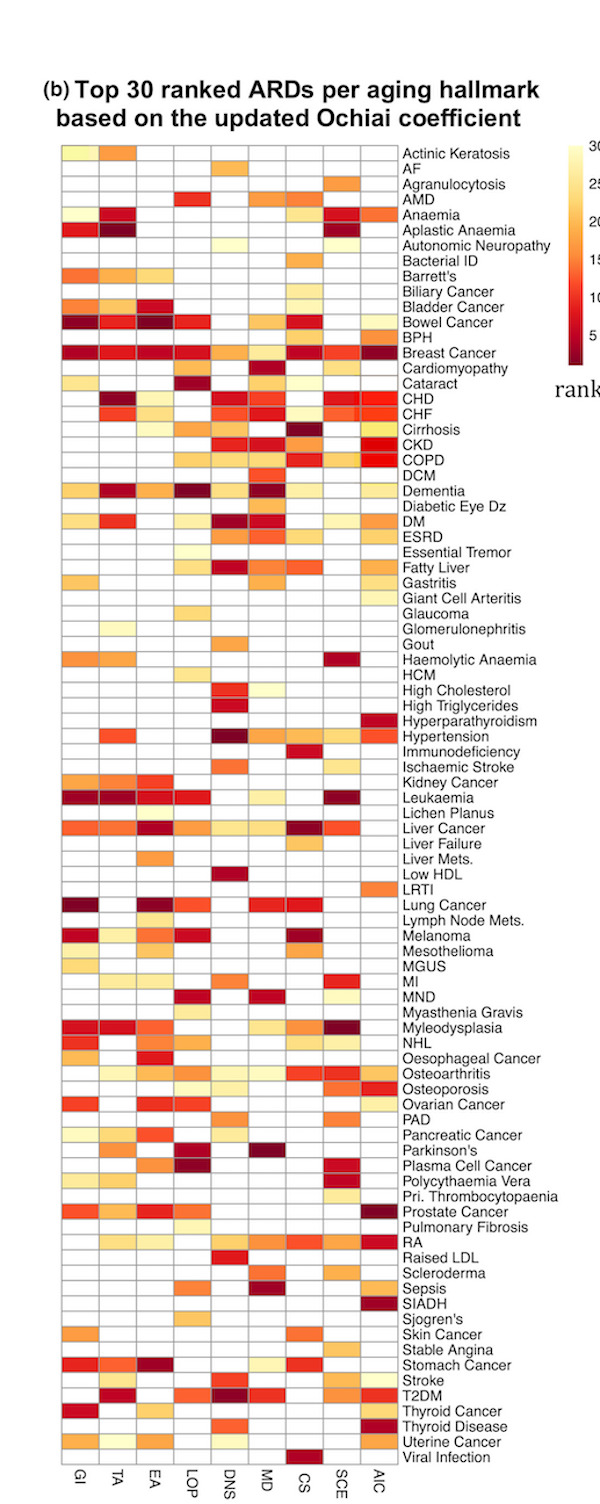
The top 30 ranked ARDs for each aging hallmark. 1st (dark red) to 30th (light yellow) ranked ARDs for a given aging hallmark are highlighted. ARDs not ranked in the top 30 are shown in white. (Click to enlarge.)
Note that the above graphic shows more than 30 ARDs, so, frankly, I’m unclear about the title, and all nine of the Hallmarks are presented even though just five are most strongly associated with ARDs. That said, let’s focus on any of the nine Hallmarks strongly associated with the most prevalent and damaging ARDS in the U.S. and much of the industrialized world:
It would be useful to understand how relevant are the associations between any Hallmark and each of the seven top diseases that kill us. To glean that, you would need to locate each disease in the table above and check its rank; meaning, the strength of the association, dark red being the strongest, and light yellow the weakest. You would also need to identify the abbreviations at the bottom of the table.
Abbreviations from the 30 top ARDs table above:
- GI = Genomic Instability
- TA = Telomere Attrition
- EA = Epigenetic Alterations
- LOP = Loss of Proteostasis
- DNS = Deregulated Nutrient Sensing
- MD = Mitochondrial Dysfunction
- CS = Cellular Senescence
- SCE = Stem Cell Exhaustion
- AIC = Altered Intercellular Communication
Strength of association between seven top diseases and Hallmarks:
- Heart Disease (CHD/CHF) — Strongest associations with:
- Telomere attrition
- Deregulated nutrient sensing
- Mitochondrial dysfunction
- Stem cell exhaustion
- Altered intercellular communication
- Cancer
- Bladder — Strongest associations with:
- Genomic instability
- Epigenetic alterations
- Bowel — Strongest associations with:
- Genomic instability
- Telomere attrition
- Loss of proteotasis
- Cellular senescence
- Breast — Strongest associations with:
- Genomic instability
- Telomere attrition
- Epigenetic alterations
- Loss of proteostasis
- Cellular senescence
- Stem cell exhaustion
- Altered intercellular communication
- Bladder — Strongest associations with:
- Lung (COPD) Strongest associations with:
- Cellular senescence
- Altered intercellular communication
- Stroke — Strongest associations with:
- Deregulated nutrient sensing
- Alzheimer’s was not indicated, but Dementia — Strongest associations with:
- Telomere attrition
- Loss of proteotasis
- Mitochondrial Dysfunction
- Diabetes (DM) — Strongest associations with
- Telomere attrition
- Deregulated nutrient sensing
- Mitochondrial Dysfunction
- Chronic Kidney (CKD) — Strongest associations with:
- Deregulated nutrient sensing
- Mitochondrial Dysfunction
- Altered intercellular communication
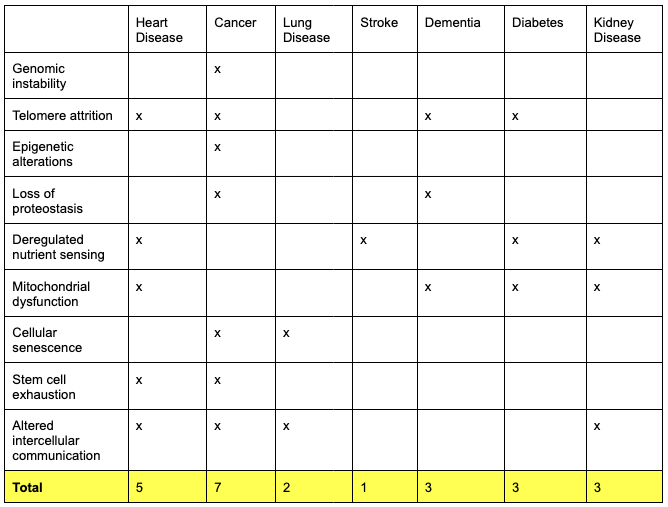
This table tallies the number of Hallmarks associated with each of the most common age-related diseases:
Thus:
- Cancer is connected to the most Hallmarks: 7
- Heart disease is next: 5
- Dementia, diabetes and kidney diseases: 3
- Lung disease: 2
- Stroke: 1
OK, so what’s the value in knowing all this?
It’s to get ahead of the diseases that kill most of us. Despite what the obituary might say, we really don’t die from “old age”, but from some disease associated with old age. What we want to know is how to prevent getting any of these diseases by finding therapies that target the aging Hallmarks strongly associated with them.
Interventions that Target Aging Hallmarks
Overall, the findings from the study we’ve been reviewing indicate that therapies, or interventions, that target aging Hallmarks may help prevent their associated age-related diseases.
Two interventions named in the study are:
- Senolytics, and
- Rapamycin.
So, according to the study we’ve been dissecting, consuming senolytics and rapamycin would be the two most effective ways to reduce your chances of getting any of the age-related diseases the study covers, which include the most prevalent in many industrial societies.
Senolytics and Rapamycin
Senolytics and senescence-associated secretory phenotype (SASP) modulators eliminate senescent cells and inhibit the SASP that harm nearby healthy cells, thereby accelerating aging. In doing this, senolytics target the Cellular senescence Hallmark, and has been proven to improve tissue health during aging and increase lifespan in mice [1] and may prevent cellular senescence-associated age-related disease multimorbidities [2].
For more about senolytics, read my post Can New Senolytic Drugs Delay Aging
Rapamycin and related compounds inhibit the mTORC1 complex in the nutrient-sensing network. mTOR inhibitors are currently the only known pharmacological intervention that increases lifespan in all experimental animal models tested. This raises the prospect that someday it might be possible to therapeutically increase lifespan, slow aging, or decrease aging-related diseases in humans [3].
The problem is that rapamycin can be a dangerous drug, as it can substantially reduce your immune response to infection. Some people are experimenting with low doses given intermittently, but the safe dose and intervals have not been established.
Given that your doctor will almost certainly not prescribe rapamycin to you for some purported longevity benefit, what else could you do to inhibit mTOR1?
The answer to that is next.
For more about rapamycin, read The Geroprotectors: 7 Anti-aging Interventions Backed By Science
Interventions that May Inhibit mTOR
mTOR is a nutrient-sensing pathway. Nutrient sensing is a cell’s ability to recognize and respond to fuel substrates such as glucose, protein and fat. Each type of fuel used by the cell requires an alternate pathway of utilization and accessory molecules needed at that time [4]. Along with mTOR, there are nutrient pathways for insulin and insulin-like growth factor, sirtuins and AMPK, among others [5].
Nutrient scarcity is a selective pressure that has shaped the evolution of most cellular processes. During food abundance, nutrient-sensing pathways engage anabolism (build up of complex molecules) and storage; whereas scarcity triggers homeostatic mechanisms, such as the mobilization of internal stores through autophagy (a catabolic process — breakdown of complex molecules). Nutrient-sensing pathways are commonly deregulated in human metabolic diseases; hence the Hallmark referred to as Deregulated nutrient-sensing.
Although rapamycin is a proven compound that will downregulate mTOR, there are other ways to do it, some perhaps better than others. Let’s look at these three:
- Fasting
- Exercise
- Supplements
#1 Fasting
Fasting is the ultimate catabolic process, and as such triggers various nutrient-sensing pathways, including mTOR, which is deregulated during fasting; meaning, less mTOR happens when you’re fasting.
When mTOR is deregulated, your body can go through a process of “self-cleaning” (autophagy) and healing, which is one reason that rapamycin has shown itself to be a life-extending comp0und — it deregulates mTOR, and mTOR inhibition increases lifespan [6].
If you have trouble fasting, one thing to try is a fasting mimicking diet (FMD). If done correctly, a FMD makes your body think it’s fasting while still eating, although the calories ingested and the macro and micronutrients have to be precisely amended to “mimic” fasting biochemically.
The only clinically tested FMD that works is ProLong, which I cover extensively here: The ProLon Mimicking Diet — Get Better, Not Older. There you can watch me coach my then 88 year-old mother through the 5-day ProLon Plan. (Yeah, she looks darn good.)
#2 Exercise
The effect of exercise on mTOR is complicated: It both activates and inhibits mTOR in various body tissues. Exercise can inhibit mTOR in the fat and liver cells, but activate it in the brain, muscles, and heart. Thus exercise is thought to be a good way to get the best out of mTOR, as Dr. Doug McDuff explains in this video:
If you don’t want to wade through the entire video, this is what I think is your best takeaway from Dr. McDuff’s presentation about the impact of exercise on mTOR:
- Eating a lot (high energy intake) stimulates mTOR via insulin and IGF-1 growth factors.
- mTOR is also activated by the mechanical loading of muscle by exercise, but when that happens, the growth factors are turned off, and thereby insulin and IGF-1 are suppressed, as are inflammatory markers.
- mTOR activation in muscle tissues and suppression of pathways involved in energy storage in the liver via insulin/IGF-1 (body fat) has a negative effect on aging; ie: improves healthspan.
If you want tips about how to exercise, see my six-part series, The Functionally Fit Fast Workout.
#3 Supplements
I’ll tell you about three supplements that studies indicate might downregulate/inhibit mTOR, but I’m not confident they do. In each case, there are other studies that cast some shade on their ability to affect mTOR, sometimes because a study couldn’t replicate the result, or it was only shown to be effective in test tubes, or the amount of the compound needed was too excessive to be practical.
Nonetheless, resveratrol, curcumin and quercetin are all safe to take, and I do just that. To be safe, do consult with your doctor before you use any new compound, as it may not mix well with a pharmaceutical you may be prescribed.
Resveratrol is a polyphenol that is a powerful antioxidant that has anti-inflammatory and neuroprotective properties. It’s found in grapes (mostly the skin and seeds), berries, and red wine. Studies have shown resveratrol inhibits mTOR. If you’d like to know more about resveratrol, check out these posts. And make sure you use the “trans” form of resveratrol and consume it with dietary fat, such as yogurt.
Curcumin, found in turmeric, may have anticancer properties through the blocking of mTOR pathways in tumor cells. When taken with piperine (black pepper) to improve bioavailabilty, it significantly helps the body decrease inflammation and oxidative stress (one of the triggers that may cause increased mTOR).
Quercetin (a natural pigment found in fruits, vegetables, and grains) has antiviral, anti-inflammatory, and anti-carcinogenic properties. It is sometimes used to supplement cancer treatment and various other diseases through its ability to inhibit mTOR activity in multiple pathways.
Your Takeaway
I recognize the complexities of understanding the Hallmarks of Aging and their associations to age-related diseases. This is something I’m endeavoring to make more clear in the anti-aging course I’m working on, as well as naming over-the-counter compounds that might be helpful to arrest the cascade between Hallmarks and downstream age-related diseases.
Of course, I could have said at the outset that what you need to do to tackle the age-related diseases predicted by the Hallmarks of Aging that will put most of us in the coffin is to:
- Fast,
- Exercise, and
- Take some supplements, maybe.
But without knowing why, why would you bother?
Regarding the “why”, remember these six things:
- The nine Hallmarks of Aging is currently considered the best overall explanation of how aging happens.
- These Hallmarks represent sources of damage, compensatory responses to damage, resulting in dysfunctional damage to us, which presents itself as all the various things we associate with aging.
- Some of these Hallmarks are strongly associated with the eventual diseases that most of us will have to contend with as we get older.
- Determining which therapies can arrest the association between Hallmarks and age-related diseases may protect us from experiencing these diseases and extend our healthspan, if not our lifespan.
- Rapamycin has had the best results at improving healthspan and lifespan in all animal models tested, which so far does not include humans. This is because rapamycin inhibits mTOR, and although you do not want mTOR to always be inhibited, it’s important that it is from time to time so that your body can heal itself through mechanisms such as autophagy. Despite rapamycin’s ability to inhibit mTOR, it can be dangerous, as the safe and effective dose for humans is presently unknown.
- Various senolytics have been shown to reduce populations of senescent cells in many studies, and there are some that are both thought to be safe and available over-the-counter, which you can read about here. Senescent cells become increasingly numerous as we age and are a major cause of inflammaging, which is very pro-aging. Ideally, to slow down the rate of aging, you want to minimize the number of senescent cells.
Get my Sunday Newsletter and be alerted when my anti-aging course is launched.
Last Updated on July 7, 2023 by Joe Garma