Can The ProLon FMD Reverse Disease and Extend Lifespan? Dr. Mark Hyman Interviews Dr. Valter Longo

The one sure way of increasing your healthy lifespan is by restricting your calories, and the ProLon FMD and other “Fasting Mimicking Diets” can get the job done without the pain. Watch Dr. Mark Hyman interview renown aging expert, Dr. Valter Longo.

IF YOU’RE interested in optimizing your healthy lifespan, you’re are in for a treat as Dr. Mark Hyman interviews Dr. Valter Longo, the inventor of the ProLon FMD, short for “Fasting Mimicking Diet”.
 Valter Longo, PhD, is the Edna Jones Professor in Gerontology and Professor in Biological Science. He is also the Director of the USC Longevity Institute.
Valter Longo, PhD, is the Edna Jones Professor in Gerontology and Professor in Biological Science. He is also the Director of the USC Longevity Institute.
He’s interested in understanding the fundamental mechanisms of aging in yeast, mice and humans by using genetics and biochemistry techniques. He’s also interested in identifying the molecular pathways conserved from simple organisms to humans that can be modulated to protect against multiple stresses and treat or prevent cancer, Alzheimer’s Disease and other diseases of aging. (1) Dr. Longo is also the author of The Longevity Diet and the inventor of ProLon FMD, a highly effective “Fasting Mimicking Diet”.
Mark Hyman, M.D. is a practicing family physician, a ten-time #1 New York Times bestselling author, and an  internationally recognized leader, speaker, educator, and advocate in his field. He’s the Director the Cleveland Clinic Center for Functional Medicine. He’s also the founder and medical director of The UltraWellness Center, chairman of the board of the Institute for Functional Medicine, a medical editor of The Huffington Post, and was a regular medical contributor on many television shows including CBS This Morning, Today Show, Good Morning America, CNN, and The View, Katie and The Dr. Oz Show. (2)
internationally recognized leader, speaker, educator, and advocate in his field. He’s the Director the Cleveland Clinic Center for Functional Medicine. He’s also the founder and medical director of The UltraWellness Center, chairman of the board of the Institute for Functional Medicine, a medical editor of The Huffington Post, and was a regular medical contributor on many television shows including CBS This Morning, Today Show, Good Morning America, CNN, and The View, Katie and The Dr. Oz Show. (2)
In the following video, Dr. Hyman interviews Dr. Longo about the anti-aging cellular signaling pathways activated by caloric restriction, fasting, fasting mimicking diets, the keto diet, what’s the best long-term diet, and much more.
I spent many hours listening to, reorganizing and adding background information so that I could present you with a somewhat cogent review of their discussion. Don’t get bogged down by the science. Find what interests you and wade into that.
Here's what's covered:
-
- Why food is a drug that can extend life
- Protein, IGF-1 and Alzheimer’s
- A quick primer on caloric restriction and its mimic
- The Intermittent Fasting FMD method
- Is the Keto Diet a FMD method?
- Inflammation, Aging and FMD
- The ProLon FMD
- So, what’s the conclusion on carbs — good or bad?
- Dr. Longo’s ideal long-term diet
- An app to track your biological age
Let’s dig in, beginning with the video for those who rather watch than read…
Why Food Is A Drug That Can Extend Life
There’s word for it now, “Nutri-technology, which refers to treating the molecules in food as drugs.
In his interview with Dr. Valter Longo, Dr. Mark Hyman points out that we’ve got 20,000 genes with five million variations in those genes, not only are they all affected by food, but so is our microbiome
Your microbiome, those microorganisms on and in you, have a hundred times more genes as those that are you’re own. They are also profoundly affected by the food you eat. When you add it all together, says Hyman, there are chemical reactions happening every second in your body; in fact, about 37 billion, billion (that’s 21 zeros) reactions in the body every second — and they’re all interacting with food.
You can get a sense of just a bit of this complexity when you look at the axis of Growth Hormone (GH), Insulin Growth Factor 1 (IGF-1), Mammalian Target of Rapamycin (mTOR), Insulin, Phosphoinositide 3-Kinase (PI3K) and Adenosine 5′ Monophosphate-activated Protein Kinase (AMPK).
Yes, this is alphabet soup of complex terminology that refers to even more complex biochemistry that the most devoted and literate biologist among you can read about in the Biogerontology paper entitled, Growth factor, energy and nutrient sensing signalling pathways in metabolic ageing, which posits:
“… metabolic pathologies are thought to be one of the main factors limiting the potential for lifespan extension.”
The principal classes of “metabolic pathologies” include these:
- Acid-base imbalance.
- Metabolic brain diseases.
- Disorders of calcium metabolism.
- DNA repair-deficiency disorders.
- Glucose metabolism disorders.
- Hyperlactatemia.
- Iron metabolism disorders.
- Lipid metabolism disorders.
And that’s not half of it. Metabolism is complicated and intertwined with much of what makes us tick.
You can visualize the “axis” I referred to above as it relates to downstream cellular signalling pathways that are activated by calorie restriction.

(Pointed arrows indicate activatory and blunt arrows inhibitory actions.)
As described in the Biogerontology paper cited above, cellular signalling pathways are implicated in age-related metabolic decline. Calorie restriction is the most potent environmental intervention that improves the metabolic profile and extends healthspan and lifespan of various animal species.
Calorie restriction is thought to be:
- Suppressing the GH/IGF-1, insulin/PI3K, and mTOR pathways, and
- Activating the AMPK pathway.
These pathways offer potential targets for therapeutic interventions to improve our metabolic homeostasis (balance) as we age — all seemingly to steam from nutrients/calories, which is a good thing, because it implies that much of this is within our control.
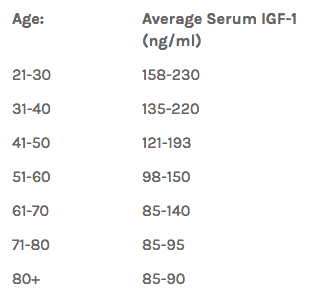
The axis indicates how these multiple cellular pathways that are linked together. All of these decline with age. For instance, at age 25, IGF-1 is high; at 90, very low, and there are times in our lives when we want it high or low.
As Dr. Joel Fuhrman puts it, “The relationship between IGF-1 levels and health is somewhat complicated.”
Excessively low or high IGF-1 levels could lead to health problems. In adults, a high IGF-1 level is linked to accelerated aging and an increased risk of cancer and premature death. Maintaining a relatively low IGF-1 level throughout most of one’s adult life is thought to be an important factor by which centenarians are able to live that long without developing cancer.
However, low IGF-1 in the elderly is linked to frailty and disease risk as well. Adequate IGF-1 levels are required for maintenance of bone mass, muscle mass and brain function at later ages.
So to prolong our lifespan, the goal is to keep up a relatively low IGF-1 throughout most of our adult life, and then as we get into our eighties and beyond, to consume enough protein so that our IGF-1 level does not get excessively low.
It is important to pay attention to our diet, to make sure our IGF-1 levels are favorable throughout life.
IGF-1 responds to dietary protein like this:
- High protein — high IGF-1, high mTOR
- Low protein — low IGF-1, high mTOR
In people 65 years old or older, no significant difference is observed in IGF-1 or mTOR in response to the level of dietary protein consumed.
Dr. longo says that once you get into your sixties and beyond, it’s a good idea to measure your IGF-1, which is a good proxy for Growth Hormone (GH) and easier to measure, as it stays stable in your blood, as opposed to GH. His example: If you’re, say, 72 years old with an IGF-1 of 280, you should lower your protein consumption; otherwise, you’re your risk for cancer increases significantly (including prostrate cancer).
Get IGF-1 tested for $57
The Life Extension Foundation works with thousands of blood labs throughout the USA. Check out their IGF-1 test here. (Note: it’s also called “Somatomedin C”.)
Dr. Fuhrman’s Ideal IFG-1 Levels
Protein, IGF-1 and Alzheimer’s
Dr. Longo has extensively studied the effects of protein on the cognitive function of mice. Yes, I realize that if you’re reading this it’s unlikely you’re a mouse, but the biology of mice are similar enough to humans that much of how they react to various experiments can be fairly accurately applied to us.
Why are mice good models for humans? Click here to find out.
The Jackson Laboratory has a good explanation for the applicability of mice models to humans.Suffice to say that nearly all of the genes in mice share functions with the genes in humans. We develop in the same way from egg and sperm, and have the same kinds of organs (heart, brain, lungs, kidneys, etc.) as well as similar circulatory, reproductive, digestive, hormonal and nervous systems.This genetic similarity also means that mice and humans inherit traits in the same way. This includes physical traits such as hair color (coat color in mice) and susceptibility to diseases such as heart disease or Alzheimer’s.These similarities make it possible for scientists to study the physiology of mice to glean information about how human beings grow, develop diseases and age. Not to mention that mice live only about two years and can be manipulated and controlled for scientific experiments.
To study the effect of dietary protein on mice, Dr Longo and his associates study mice that had human mutations that made them develop the mouse equivalent of Alzheimer’s. They presented all the symptoms of Alzheimer’s, including various cognitive impairments.
The scientists altered the IGF-1 of these mice up and down through increasing and decreasing the quantity of protein (the “essential” amino acids needed for normal function) fed to the mice. What they determined was that they could improve the cognitive capacity of the mice as they got older by increasing the amount of protein ingested.
Now, it’s important to know that IGF-1 is a double-edged sword that can cut both ways. For the elderly prone to, or experiencing cognitive issues want their IGF-1 to be low, but people of the same age who may be experiencing recovery from an acute neuronal insult, such as a stroke, is augmented in the setting of higher IGF-1. (3)
Now that you have a sense about some of the science about caloric restriction and the cellular signaling pathways it influences, let’s turn what is meant by caloric restriction, and the various ways to do it.
A Quick Primer On Caloric Restriction and Its Mimic
“Caloric Restriction” (CR) used to exclusively mean eating 30% fewer calories all the time. The mice and monkeys that have been extensively studied in CR research can’t complain, but the people who tried can, and they make it clear that on CR they’re frustrated, irritable and hungry.
But it works.
To state the obvious, you lose weight when restricting calories below your metabolic requirement. What’s not so obvious are the health benefits.
CR and Fasting Mimicking Diets Have Been Shown to Help:
- Decrease Weight and Body Fat
- Stem Cell Regeneration
- Keep Glucose at Healthy Levels
- Keep CRP at Healthy Levels
- Keep Triglycerides at Healthy Levels
- Keep Blood Pressure at Healthy Levels
- Keep Healthy Levels of Bone Density
- Impact in IGF-1 (Aging Marker)
- Maintain Insulin Resistance at Healthy Levels
- Maintain Cholesterol at Healthy Levels
- Decrease Visceral Fat
That’s all great stuff — but is it possible to “mimic” CR so that you still get the benefits?
Yes, indeed, Fasting Mimicking Diets, if properly constructed, produce the same favorable health markers as CR. Fasting Mimicking Diets, such as Dr. Longo’s ProLon FMD, lowers insulin, glucose, IGF-1, leptin, inflammation and more. It would take many drugs to attain same effects.
It makes sense that this is true, because this is how we evolved. All animals – including humans back in the day — have periods of eating and fasting. That was Dr. Longo’s insight. He began experiments with mice where he put them thru periods of water-only fasting and then eating. He discovered that this made them resistant to cancer.
Unfortunately, we humans don’t want to fast. Cancer patients, in particular, are more resistant to fasting than those without the disease, which thwarted Dr. Longo’s attempts to administer a fasting program for them. What he did instead was to develop the ProLon Fasting Mimicking Diet, or ProLon FMD. It “mimicked” fasting in that this diet led to the same health benefits that short-term fasting did. Moreover, Longo’s ProLon FMD proved to be safe and patients were compliant; meaning, they stayed on the diet to get the benefits.
What basically happens on the ProLon FMD is that it activates the body’s own capacity to heal.
An example of the effectiveness of this FMD approach is immunotherapy.
The 2018 Nobel Prize in Physiology or Medicine was awarded jointly to two cancer immunotherapy researchers, James P. Allison, PhD, of The University of Texas MD Anderson Cancer Center, and Dr. Tasuku Honjo of Kyoto University in Japan for their work on uncovering ways to activate the immune system to attack cancer, and only cancer, a breakthrough in developing new cancer treatments. (4)
Dr. Longo discovered that using immunotherapy along with fasting, or a FMD, provides a beneficial synergistic effect.
The lesson here is that restricting or eliminating calories from time to time enables our bodies to heal.
The Intermittent Fasting FMD Method
The term “intermittent” doesn’t convey much. The idea is that you restrict calories or have none (complete fast) for some number of hours. To have some, but minor, beneficial effects for weight loss or autophagy, the least is 12 hours. The target is typically 16 hours of fasting, leaving eight hours for feeding.
Dr. Sachin Panda has an ongoing self-reporting study where people simply log when they eat, not what they eat. After examining thousands of data points from participants logging what they eat, Dr. Panda has found that most people eat five to six times during a 15 hour period; thus, only nine hours of fasting. What is desired is the inverse of that. But not right away (you must ease into it), and not for sustained periods of time.
Longo recommends that you begin with 12/12 — 12 hours of fasting and 12 feeding. Gradually you can extend the fasting period for an hour or two, and cycle in and out of a week or so each month where you’re fasting for 16 hours, feeding for eight. This is safe.
Read my posts:
Is The Keto Diet A FMD Method?

Typical keto breakfast
The keto diet w/ protein or sugar limit FMD effect. If Keto high fat but no veggies (carbs — 45% in FMD, 45% FAT, 10% protein). The benefit of veggies over course of many cycles is to keep up the carb ingestion during normal diet. Don’t want back and forth between high fat and high carbs.
Th keto diet can be useful for obese people, but not long-term, says Dr. Longo. If you’re going to do keto, he recommends 80% fat, 10% veggie/legume carbs and 10% protein also legumes (lentils are high in protein) and fatty fish.
The epidemiological data clearly shows, he says, that animal-based low carb diets can be harmful long-term. Here he recommends that people on a keto diet choose predominately vegetable-based fats (avocados, olive oil, flax seeds/oil and hemp seeds/oil) over animal based fats (diary, meat).
Could The Ketogenic Diet Help With Cognitive Decline?
Dr. Longo and his team are actively working with early-onstage Alzheimer patients using his ProLon Fasting Mimicking Diet as a therapy. (It is, however, available to the public.) The protocol in this case is to have these patients do the five-day ProLon diet only once every two months, because of the concern about frailty and muscle loss. In addition to the ProLon Fasting Mimicking Diet, these patients are given a ketogenic supplement for one month to help make sure they don’t lose weight.
Dr. Hyman points out that the ketogenic supplement is important, because Alzheimer’s is like a “type 3” diabetes that occurs in the brain in that it can’t use glucose/sugar very well but can use fat or ketones. Hyman has put his cognitively-challenged patients on a ketogenic diet and they improved.
Both doctors agreed that the older you are, the more careful a transition to extreme diets need to be; therefore, elderly folk interested in trying a ketogenic diet should gradually ease into it and be monitored by their doctor.
In addition, as Dr. Longo stresses, it’s not known if such a diet is beneficial for long-term use, something I explore in my article, The Solution To The Keto Diet Controversy. Dr. Hyman relented to Longo’s point about safety, but expressed his experience with his patients is that they generally do well on the keto diet. (The debate begins at about time stamp 50:00.)
One major benefit of a FMD that does not occur to the same degree with keto is the benefical effects on gut bacteria. The prebiotic ingredients in a FMD that is vegetable-dominant feed the “good bacteria” in our guts, such as lactobacillus bifidobacteria. This has the effect of reducing inflammation (which would not result from either from a keto FMD or water only fast), a major problem we grapple with as we age.
Inflammation, Aging and FMD
Note that we’re speaking about chronic inflammation, not the kind that temporarily happens — called acute inflammation — when you cut on your hand. In this case, chemicals known as cytokines are released by the damaged tissue. The cytokines act as “emergency signals” that bring in your body’s immune cells, hormones and nutrients to fix the problem.
This is protective and useful.
In contrast to acute, inflammation, chronic inflammation associated with aging happens due to cellular dysfunction. As we age, “junk” accumulates inside of our cells, such as DNA mutations, damaged mitochondria, protein accumulation (such as beta amyloids in the brain, thought to be a cause of Alzheimer’s). Our immune system perceives this as foreign invaders and attack them, thus beginning the process of inflaming the body and keeping it in that state.
The ProLon FMD
Dr. Longo’s ProLon Fasting Mimicking Diet (ProLon FMD) is a five-day dietary program that nourishes your body while promoting regenerative and rejuvenating changes, including supporting healthy levels of a range of physiological markers that contribute to aging, such as cholesterol, inflammation, and fasting glucose.
In experiments on mice on the FMD, Longo and his team damage their pancreas so they stop making insulin. Then the mice are fed a ProLon FMD-type diet and in response, the pancreas turns on an embryonic developmental program, such that all the genes that were “turned down” when the pancreas is first generated when the mouse was born now get “turned up”. Clearly, says Dr. Longo, the fasting mimicking diet has activated a program, not simply a few gene, but all of them.
When you cut your skin, it is not repaired by chance. Every cytokine (a large group of proteins, peptides or glycoproteins that are secreted by specific cells of immune system) and stem cell gets recruited and knows exactly where to go, and slowly the cut is repaired.
Likewise, the ProLon FMD can trigger various programs in the liver, pancreas, metabolism, brain, etc. Longo believes this happens because of a “strongly evolved program” that has to do with the availability of food. When there’s no food, the body has evolved an ability to use body fat storage to meet metabolic requirements.
This regenerative capacity has clearly been demonstrated, Longo says, in people who can regrow emaciated organs and muscle after nearly starving to death once food is consistently available to them. In this situation, stem cells, and other cells, respond to incoming nutrients and regenerate the body.
Read my article, The ProLon Fasting Mimicking Diet — Get Better, Not Older.
So, What’s The Conclusion On Carbs — Good or Bad?

Dr. Hyman steered the conversation to the high carb/high fat debate at time stamp 55:59. He said he follows the work of Dr. David Ludwig, a Harvard professor renown for his opposition to sugar and high glycemic carbohydrates (carbs that typically boost blood sugar).
Dr. Ludwig, says Hyman, did a $12 million study published on BMJ that looked at the effect of carbohydrates on metabolism; specifically, to determine the effects of diets varying in carbohydrate to fat ratio on total energy expenditure.
The study compared the effects of low carb/high fat and low fat/high carb diets on 164 adults aged 18-65 years with a body mass index (BMI) of 25 or higher (the overweight range). Because the aim was to study energy expenditure/metabolism, not weight loss, all participants had a caloric intake designed to limit their weight loss to 2 kg (4.4 lbs). Protein intake was controlled. What was varied was the proportion of carbs consumed. The test lasted for 20 weeks.
Participants were randomly assigned to one of three test diets according to carbohydrate content:
- High carbs, 60% of total macronutrients (fat, carbs and protein)
- Moderate carbs, 54% of total macronutrients
- Low carbs, 20% of total macronutrients
The studies conclusion:
“Consistent with the carbohydrate-insulin model, lowering dietary carbohydrate increased energy expenditure during weight loss maintenance. This metabolic effect may improve the success of obesity treatment, especially among those with high insulin secretion.” (5)
As he said to Dr. Longo, Dr. Hyman’s takeaway from the study is that:
“… the ones who had the high fat diet had much faster metabolisms. In other words they burned an extra 250 calories a day, and if they were insulin resistant they’d burn an extra 400 calories or 450 calories a day. And that would lead me to believe that the carbohydrates that they were eating are stimulating insulin, which is really, I think, one of the main drivers of aging — activating insulin and activating all the inflammatory pathways that go with it [insulin resistance affecting the brain, cancer and heart disease].” Ending with, “how do you explain this… the data just seems all over the place?”
To which Dr. Longo basically replies, “Not so fast.”
Longo’s response to Hyman’s assertion is that the data is not all over the place. He tells Hyman that the data seems like it’s all over the place till you look at epidemiology clinical studies, basic research and studies of centenarians.
In mouse studies, says Longo, a high fat diet does make them lose weight and look good, but they die earlier than the other mice. He says that very low lean body mass can be good when you’re younger but that some fat storage is beneficial as you get to 70 years and beyond, because fat stores stem cells, and protects organs from stress and movement.
Basically, Longo is underscoring that there’s plenty of nuance to the effects of diet on health. For instance, he points out that studies on mice show that caloric restriction isn’t necessarily some panacea for health and longevity, citing that studies show that one-third of mice calorically restricted live longer, but that one-third have neutral effects and one-third have shorter lifespans. And the group of mice that had the shortest lifespans were those with the lowest fat storage.
Longo presents the duality like this:
- On one side you have central adiposity (obesity) visceral fat (fat stored within the abdominal cavity) and liver fat driving various diseases.
- One the other side, subcutaneous fat (fat under the skin) and other fat could be helping an individual — at least in mouse models — live longer.
Agreeing with Dr. Hyman about the dangers of excess carbohydrates, Dr. Longo admits that “eventually” a diet consisting of plentiful high glycemic carbs like pasta, white bread and sugary fruits (like, say, bananas, not berries) will cause a problem, but he points out situations where the right kind of carb-dominant diets have great health outcomes, such as with the famously long-lived Japanese Okinawans.
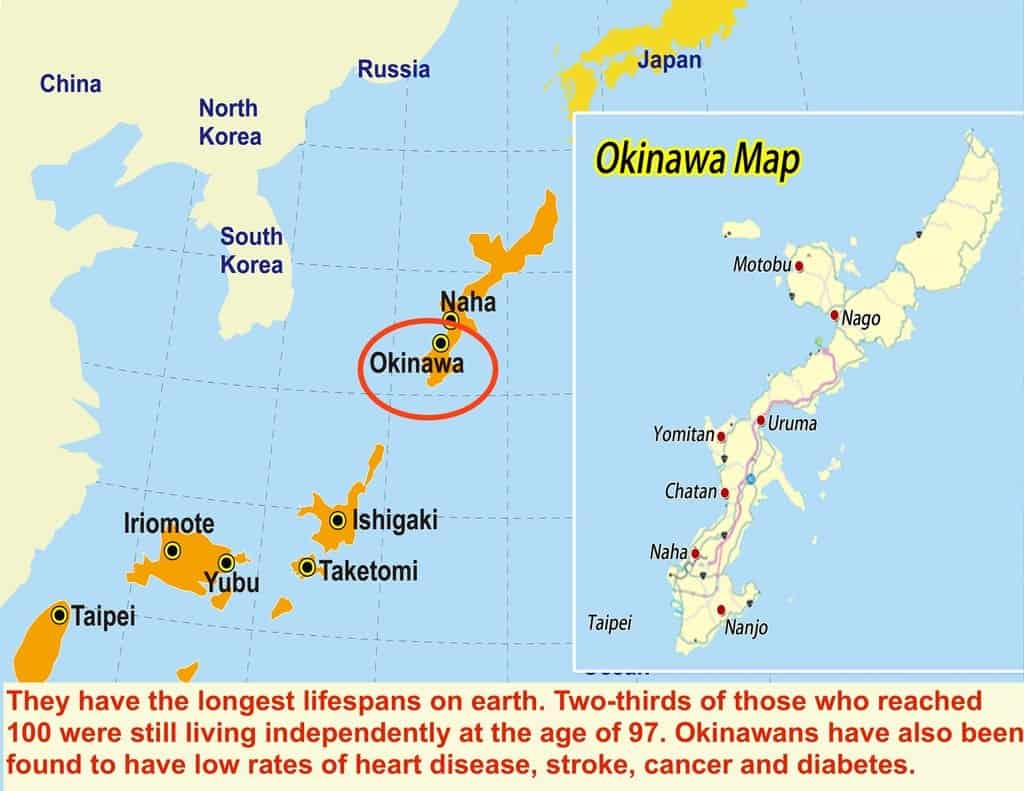
Common characteristics of the Okinawan Diet and lifestyle:
- Low-calorie, high-carbohydrate ― mainly due to the sweet potatoes (despite their name, a low glycemic carb with a modest effect on blood sugar) ― moderate-protein and nutrient-rich diet, with sparse amounts of meat, refined grains, sugar and dairy.
- Small amount of protein — the ratio of protein (mostly fish) to carbohydrates in the Okinawan diet is 1-to-10.
- High in fiber and fermented foods.
- Practice what is called hara hachi bu, which means eat until you are 80% full.
- Socially engaged.
- Active.
- Elderly are not fat, but have a bit of excess fat.
Dr. Longo points out that until in recent decades, people in his native Italy had a similar diet and lifestyle to the Okinawans, and in various parts of Italy where this was true lived some of the healthiest and longest lived people on the planet. What happened recently, however, he explains is that people went from consuming good carbs to bad carbs. When that happened, obesity rates skyrocketed, people became unhealthy and died younger.
In this situation — eating too many unhealthy, processed, high glycemic carbs, the ketogenic diet might be useful, admits Longo, which is Dr. Hyman’s contention. The question at hand is, for how long should you be on the keto diet, posits Longo.
Longo and Hyman Agree — If obese, cycle between keto and healthy carbs
We finally get to the bottom line, one that both Drs. Longo and Hyman agree.
To sum it up, it’s this:
If you’re overweight or obese, you can use the keto diet for a couple of months at time with the goal to end up on a long-term longevity diet, such as that practised by the Okinawans.
Longo stresses that if your diet is carb-dominant, there must be a focus on the “good carbs”; namely:
- Vegetables, and
- Legumes (beans, lentils, chickpeas)
(Note: If you’re concerned about the so-called “anti-nutrients” claimed to be in legumes, such as lectins and phytates, simply soak them in water overnight before cooking. You can also make them more nutritious by letting sprout slightly before cooking.)
Dr. Valter Longo’s Ideal Long-term Diet
Longo thinks the ideal long-term diet has these macronturient proportions:
- 60% from carbs, mostly from vegetables and legumes
- 30% fats mostly from olive oil, nuts and seeds and a fatty fish yeah and
- 10% protein mostly from legumes and fatty fish
He says he came to this conclusion from going around the world looking at the basic research, looking at the clinical trials and observing the diets of the longest lived people.
I think gorrillas would agree — Read Eat Like A Gorilla — Your Scientifically Proven Best Diet.
Coming Soon — An App to Track Your Biological Age
Right now, you’d have to jump through a lot of hoops to get a sense for how well you’re aging biologically, as opposed to chronologically. Your chronological age is obvious, right — just subtract today’s date from when you were born. But that number may say little about the age of your physiology, your biology.
All of us know people who seem younger and older than their chronological age. Why is that? Assuming that how they look on the outside mirrors how they, in effect, look inside insides, such “youngsters” are aging biologically slower, or older, than is typical.
How do you find out how you’re aging?
Different studies come up with a group of somewhat different biometrics, such as IGF-1, glucose, lean body mass, LDL, etc. The list can be quite large, and each metric can have a disproportionate effect on aging.
Given this, I was heartened to learn that Dr. Longo and his team will soon release an app to approximate biological age.
At time stamp 69:39, Dr. Hyman asks Dr. Longo how to track biological aging. Dr. Longo’s response:
“L-Nutra, the company that I founded [the maker of the ProLong Fasting Mimicking Diet] is going to have an app soon. This is something that Morgan Levine from Yale [School of Medicine] came up with. There’s ten different markers… that are very good predictors of mortality… What we’re looking at are things like cholesterol, c-reactive protein [a measure of chronic inflammation], [perhaps] IGF-1… many markers, [perhaps] nine or ten.”
Longo further explains that the app will enable you to enter your “numbers” obtained by various tests of these markers, and it will spit out your biological age — the age your body actually is.
Although he doesn’t disregard more esoteric measures of biological aging, such as telomere length or some ideal proportion of senescent cells to healthy cells, Longo emphasizes that we can more easily find out how we’re aging by assessing the basic hallmarks of aging, such as chronic inflammation, white blood cell count, IGF-1, insulin resistance and cognitive function.
Your Takeaway
There’s a lot packed into this article, and it would be odd if you weren’t a bit confused by all that was thrown at you.
The good news is the bottom line is straightforward and may be encapsulated into four statements:
- Caloric restriction is a proven method to reverse many chronic diseases associated with aging, and to potentially extend healthy lifespan.
- A persistent reduction in calories makes most people miserable, as does water-only fasting, so various fasting mimicking diets (FMDs) have been tested to also offer beneficial health outcomes.
- FMDs can include intermittent fasting, the Prolon FMD and in some cases the Keto Diet, if protein ingestion is constrained and animal sources of dietary fat are minimized in favor of fats from vegetable oils, avocado, fish, nuts and seeds.
- The best way to use the Keto Diet is to cycle in and out of it. The best diet long-term is one that is dominated by plant food, fish and non-animal derived fats. The Okinawains are a good model.
The Longevity Diet: Discover the New Science Behind Stem Cell Activation and Regeneration to Slow Aging, Fight Disease, and Optimize Weight
Food: What the Heck Should I Eat?
The End of Heart Disease: The Eat to Live Plan to Prevent and Reverse Heart Disease
Last Updated on November 23, 2022 by Joe Garma






